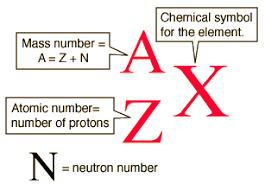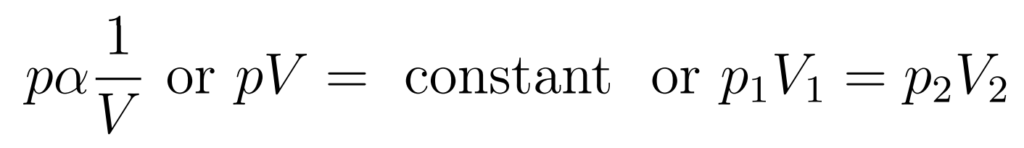Thermal Physics - Topic 3
Bite-sized Thermal Physics Study Notes for IB Physics HL/SL
Table of Contents

Key point
- Matter consists of very small particles
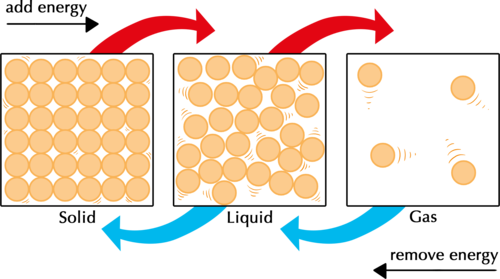
The particle model of matter states that all matter is composed of small particles.
There are three types of states:
- Solid
- Liquid
- Gas
In a solid, particles can be modeled by springs. The strings represent bonds.
- A solid has very strong bonds
- Inter-particle forces are very high
- Particles are in a regular arrangement
- Particles vibrate around a fixed position
In a liquid, particles will take the shape of the container they are placed in.
- Medium-strong bonds
- Inter-particle forces are somewhat high
- Particles are in a random arrangement
- Particles can slide past one another
Gas is the least dense state of matter.
- Gas has very weak bonds
- Inter-particle forces are very low
- Particles are in a random arrangement
- Particles are free to move in all directions
Key points
- Temperature is proportional to the random kinetic energy of a molecule
- Lowest possible temperature is the absolute zero
Temperature is a measure of ‘hotness’ or ‘coldness’ of a body.
- Proportional to the random kinetic energy of a molecule
- Kelvin Scale – to illustrate the ‘hotness’ or ‘coldness’ of a body
- Since random kinetic energy can be 0, the temperature can be 0 too – absolute zero
- The magnitude of a Kelvin is the same as that of a degree of Celsius
T (in Kelvin, K) = T (in degrees Celsuis, C) + 273
A Thermometer is used to measure temperature.
- Must possess a property that changes in a predictable way with varying temperature
- When the thermometer is in contact with the substance in question, then a thermal interaction occurs until the thermometer and the substance are in a thermal equilibrium
Key point
- Heat is energy that is transferred between bodies
Heat is the energy that is transferred from one body to another body as a result of a difference in temperature.
- Each Molecule consists of particles
- Kinetic Energy – the speed of vibration
- Potential Energy – the separation between the particles
- Internal Energy – the sum of Kinetic Energy and the inter-particle potential energy
- Heat transferred between a hot and a cold body increases the internal energy of the cold body
- Work done on particles increases the potential energy
- Thus the internal energy can change due to work performed or heat transfer
- Internal Energy, heat, and work are all measured in Joules (J)
- Temperature is a measure of the random kinetic energy of a molecule
Formula Booklet
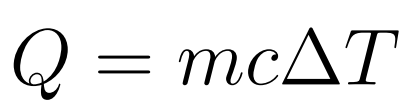
Specific Heat Capacity is the amount of energy needed in order to increase a unit of mass of the object by one kelvin.
- Q = Energy/Heat
- m = mass
- c = specific heat capacity
- ΔT = change in Temperature
Key point
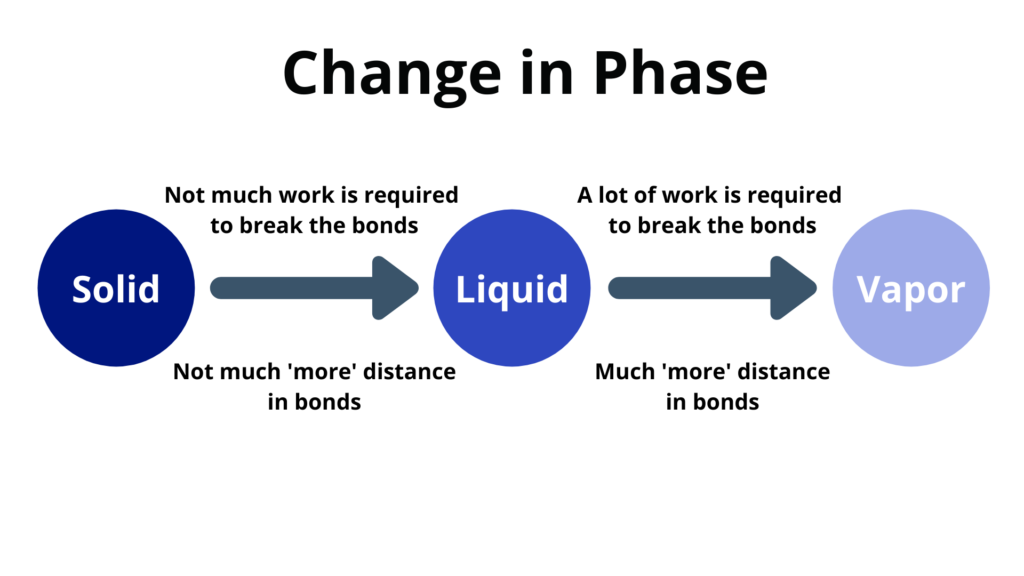
- When heat is provided to a body it doesn’t mean that the body will change its temperature – it may also change its phase
Change of phase happens at a constant temperature.
- Melting – when a solid changes to a liquid
- Freezing – when a liquid changes to a solid
- Vaporization – when a liquid changes to a vapor
- Condensation – when a vapor changes into a liquid
- The heat energy goes into the potential energy instead of the kinetic energy
Formula Booklet
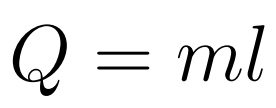
Specific Latent Heat (l) is defined as the amount of energy required to change the phase of a unit mass at a constant temperature.
- Q = Energy/Heat
- l = Specific latent heat
- m = mass
Worksheet/PPT
Key point
- Remember the fundamentals about Physics and its Thermal Concept Laws!
Thermal Concepts Physics Worksheet
Questions/Answers
Key point
- The number of particles in a mole is equal to the Avogadro Constant
- A ‘particle’ can mean an atom or a molecule
- The number of particles in a mole is N A
Formula Booklet
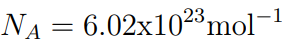
Formula Booklet
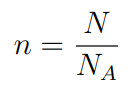
If a substance contains N particles, then the number of moles ‘n‘ is given by this formula.
- n = number of moles
- N = Number of particles
- NA = Avogadro’s Constant
The atomic mass unit (1u) is defined as 1/12 of the mass of a carbon-12 atom.
- The mass per one neutron is 1 mole
- The mass per one proton is 1 mole
- Molar mass is the sum of all atomic masses of a molecule
Formula Booklet
Unified atomic mass unit
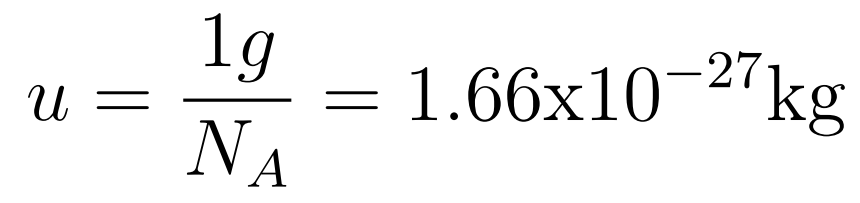
Key point
- Pressure is the normal force applied per unit area
- The force is applied normal to the area
- If the force is given by with an angle, then find the vertical component of the angle
- P = Pressure
- F = Force
- A = Area
- Unit: Newton per square metre, Nm-2 = pascal, Pa
- Can also be used: atmospere, atm = 1.013 x 105 Pa
Formula Booklet
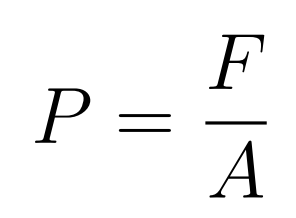
Key points
- An ideal gas is a theoretical model of a real gas
- A real gas can be approximated by an ideal gas at low density (low pressure, high temperature)
An Ideal Gas is a theoretical model of a gas. It is defined by the following assumptions:
- The molecules are point particles – with negligible volume
- The molecules obey the laws of mechanics
- An ideal gas can not be liquefied or solidified
- Molecules have a variety of speeds and move randomly
- There are no forces between the molecules except when the molecules collide
- The duration of a collision is negligible compared to the time between collisions
- The collisions of the molecules with each other and with the container walls are elastic
At very low density (low pressure, high temperature) a real gas will generally behave like an ideal gas.
Key point
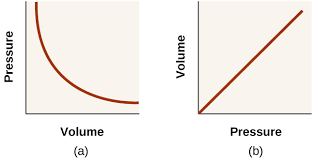
- Pressure is inversely proportional to volume
The Pressure-Volume Law states that at constant temperature and with a fixed quantity of gas, pressure is inversely proportional to volume.
- This relationship is also known as Boyle’s Law
- The curve is isothermal – the temperature at all points on the curve is the same
- The product pV is the same for all points on the curve
Key point
- Volume increases uniformly with temperature
The Volume-Temperature Law states that when temperature is expressed in kelvin, then at constant temperature volume increases uniformly with temperature.
- Charle’s Law
Not in Formula Booklet but important
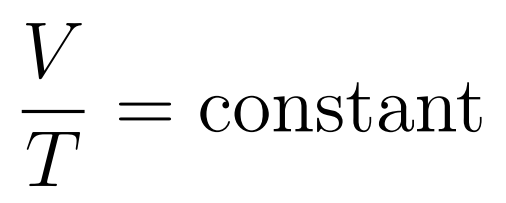
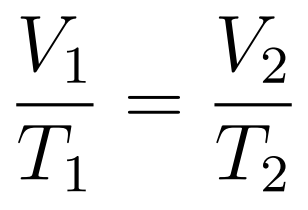
Key point
- Pressure increases uniformly with increasing temperature
The Pressure-Temperature Law states that when temperature is expressed in kelvin and volume is constant, then pressure increases uniformly with increasing temperature.
- Gay-Lussac’s Law or Amonton’s Law
Not in Formula Booklet but important
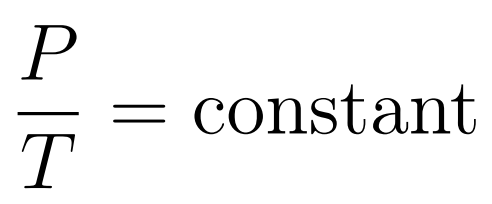
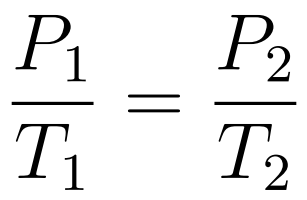
Key point
- The Gas constant (R) is a universal constant
The Equation of state of an Ideal Gas relates Volume, Pressure, Temperature, and the number of moles of a gas. .
- R is the universal gas constant
- p = pressure
- V = Volume
- n = number of moles
- T = temperature
- Through the introduction of the universal gas constant one can now calculate for the missing quantities (with the help of the given quantities)
Formula Booklet
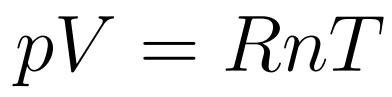
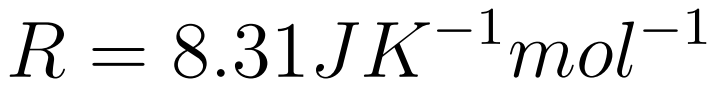
Key points
- Calculate the Kinetic Energy of a gas
- The Internal Energy of an ideal gas consists only of its kinetic energy
The molecules of a gas move randomly with a range of speeds.
- kb = Boltzmann constant
- T = Temperature
- EK = Kinetic Energy
Formula Booklet
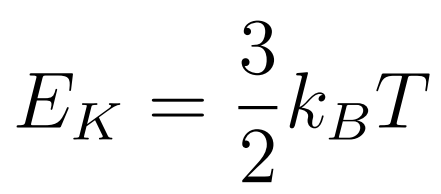
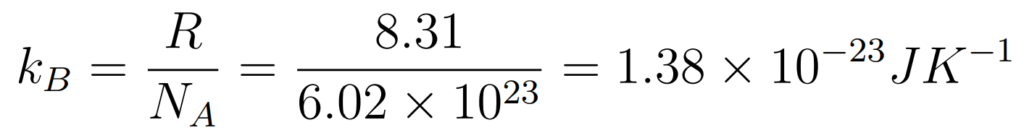
Not in Formula Booklet but important
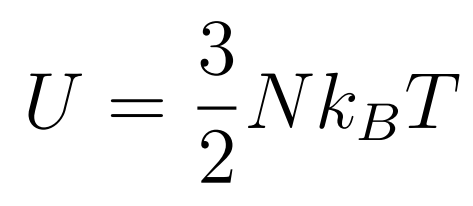
The Internal Energy of an ideal gas consists only of the random kinetic energy of its molecules
- N = Number of Molecules
- kB = Boltzmann constant
- T = Temperature
- Remember the basic rules that characterize an ideal gas
Worksheet - Kinetic Molecular Theory of Gases
Worksheet with Answers
Questions and Answers
Worksheet with Answers 2.0
Questions and Answers
PPT (PDF) - Kinetic Molecular Theory of Gases
Subscribe to the Inertia Newsletter
IB News, Covid-19 Updates, Deadlines, Tips and Tricks, and Hundreds of Free Resources are Awaiting You!
Features
- Study Notes
- Thousands of IB Questions
- Detailed Answers
- Ask-A-Question System